ZR-75-1
Target ID: CHEMBL614393
Organism: Homo sapiens
Type: CELL-LINE
Molecules
| Name | SMILES | Bioactivity | Structure |
|---|---|---|---|
| METHOTREXATE | CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(C(=O)N[C@@H](CCC(=O)O)C(=O)O)cc1 | ID50: 6.2 nM | 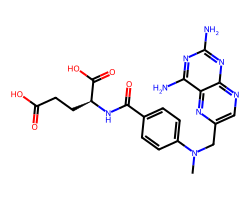 |
| ERLOTINIB | C#Cc1cccc(Nc2ncnc3cc(OCCOC)c(OCCOC)cc23)c1 | IC50: 1450.0 nM | 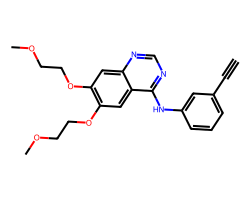 |
| VORINOSTAT | O=C(CCCCCCC(=O)Nc1ccccc1)NO | Inhibition: 100.0 % | 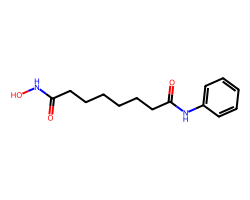 |
| EMETINE | CC[C@H]1CN2CCc3cc(OC)c(OC)cc3[C@@H]2C[C@@H]1C[C@H]1NCCc2cc(OC)c(OC)cc21 | IC50: 40.0 nM | 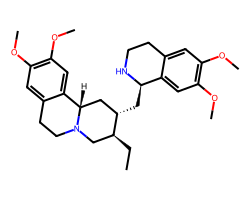 |
| BAICALEIN | O=c1cc(-c2ccccc2)oc2cc(O)c(O)c(O)c12 | Inhibition: 57.0 % | 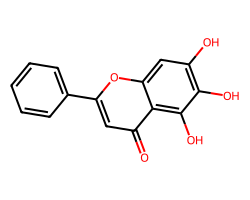 |
| FLOXURIDINE | O=c1[nH]c(=O)n([C@H]2C[C@H](O)[C@@H](CO)O2)cc1F | IC50: 2.0 nM | 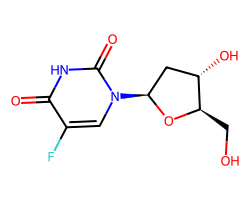 |
| SUNITINIB | CCN(CC)CCNC(=O)c1c(C)[nH]c(/C=C2\C(=O)Nc3ccc(F)cc32)c1C | IC50: 80.0 nM | 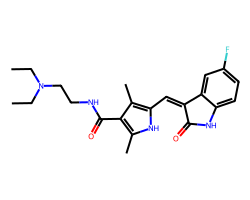 |
| CRIZOTINIB | C[C@@H](Oc1cc(-c2cnn(C3CCNCC3)c2)cnc1N)c1c(Cl)ccc(F)c1Cl | IC50: 8.0 nM | 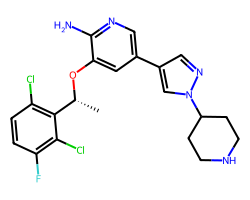 |
| IMATINIB | Cc1ccc(NC(=O)c2ccc(CN3CCN(C)CC3)cc2)cc1Nc1nccc(-c2cccnc2)n1 | IC50: 40.0 nM | 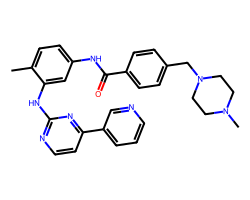 |
| TOZASERTIB | Cc1cc(Nc2cc(N3CCN(C)CC3)nc(Sc3ccc(NC(=O)C4CC4)cc3)n2)n[nH]1 | Ki: 0.6 nM | 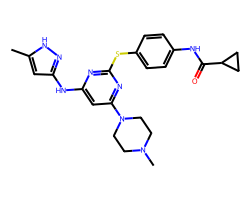 |
| TAMOXIFEN | CC/C(=C(\c1ccccc1)c1ccc(OCCN(C)C)cc1)c1ccccc1 | IC50: 6750.0 nM | 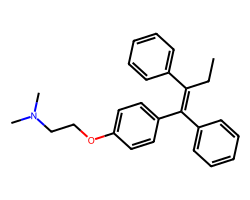 |
| SORAFENIB | CNC(=O)c1cc(Oc2ccc(NC(=O)Nc3ccc(Cl)c(C(F)(F)F)c3)cc2)ccn1 | IC50: 12.0 nM | 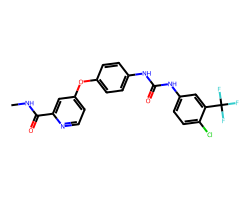 |
| SARACATINIB | CN1CCN(CCOc2cc(OC3CCOCC3)c3c(Nc4c(Cl)ccc5c4OCO5)ncnc3c2)CC1 | IC50: 2.7 nM | 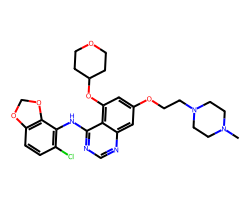 |
Top Fragments
| SMILES | Avg pKi | Count | Structure |
|---|---|---|---|
| [5*]N[5*] | 7.75 | 312 | 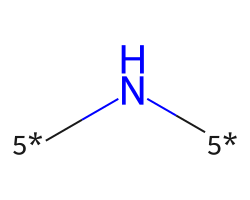 |
| [1*]C([6*])=O | 7.97 | 195 | 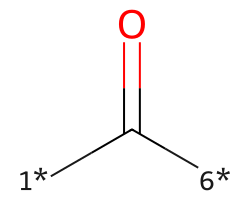 |
| [16*]c1ccc([16*])cc1 | 7.73 | 184 | 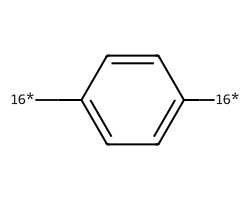 |
| [3*]O[3*] | 7.24 | 184 |  |
| [4*]CC[4*] | 6.78 | 145 |  |